Is your gut not quite right since having COVID-19? You do not have to live with these lasting effects. Rutgers scientists recently found that COVID-19 acute infection disrupts gut bacteria balance, especially with antibiotic treatment. Microbiome analysis and functional medicine available at AIMA of Nashua can help identify and address the specific bacterial imbalance causing your symptoms. Come see us to develop a plan to address the cause.
Gut microbes disturbed by COVID-19 infection, especially with antibiotics
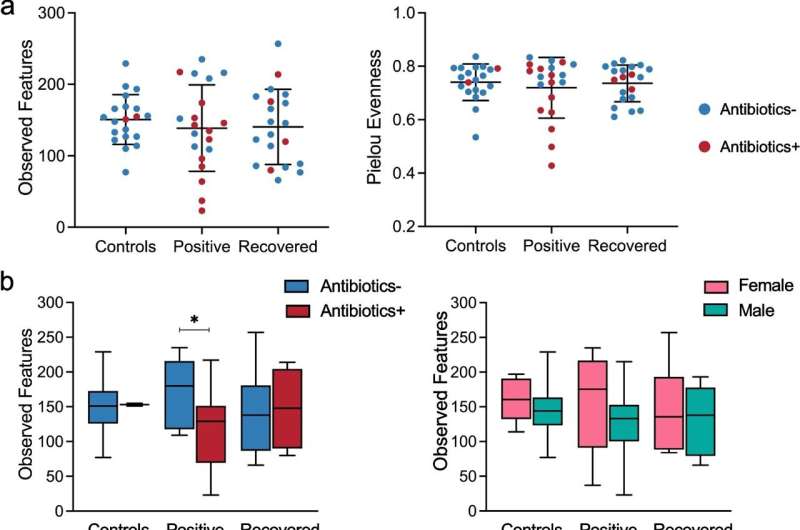
Gut microbial alpha-diversity in three groups of study subjects, based on 16S rRNA sequences in fecal samples. a Species richness and evenness of fecal samples from Controls (n = 20), COVID-19-positive patients (n = 20), and COVID-19-recovered patients (n = 20) were measured in terms of observed OTUs and Pielou evenness, respectively. Differences between groups were not significant using Welch’s t-test. b Species richness in subjects with or without antibiotic use, and in relation to sex, was estimated by observed OTUs. *p < 0.05 by Welch’s t-test. c, d Alpha-diversity in subjects without (c) or with (d) antibiotic use was compared between groups using the metrics of observed OTUs, Shannon index, and Pielou evenness. *p < 0.05 by Welch’s t-test. Credit: Molecular Biomedicine (2022). DOI: 10.1186/s43556-022-00103-1
In an intensive look at the effects of the virus causing COVID-19 on patients’ microbiome—the collection of microorganisms that live in and on the human body—Rutgers scientists found that acute infection disrupts a healthy balance between good and bad microbes in the gut, especially with antibiotic treatment. The work may lead to the development of probiotic supplements to redress any gut imbalances in future patients, the scientists said.
Reporting in the journal Molecular Biomedicine, researchers described the first results of an ongoing study examining the microbiome of patients and volunteers at Robert Wood Johnson University Hospital in New Brunswick. The study, which began in May 2020, the early days of the pandemic, was designed to zero in on the microbiome because many COVID-19 sufferers complained of gastrointestinal issues—both during the acute phases of their illness and while recuperating.
“We wanted to gain a deeper understanding by looking at specimens that would give us an indication about the state of the gut microbiome in people,” said Martin Blaser, the Henry Rutgers Chair of the Human Microbiome at Rutgers University, director of the Center for Advanced Biotechnology and Medicine (CABM) at Rutgers and an author on the study. “What we found was that, while there were differences between people who had COVID-19 and those who were not ill, the biggest difference from others was seen in those who had been administered antibiotics.”
Early in the pandemic, before the introduction of vaccines and other antiviral remedies, it was a common practice to treat COVID-19 patients with a round of antibiotics to attempt to target possible secondary infections, said Blaser, who also is a professor of medicine and pathology and laboratory medicine at Rutgers Robert Wood Johnson Medical School.
Humans carry large and diverse populations of microbes, Blaser said. These microorganisms live in the gastrointestinal tract, on the skin and in other organs, with the largest population in the colon. Scientists such as Blaser have shown over recent decades that the microbiome plays a pivotal role in human health, interacting with metabolism, the immune system and the central nervous system.
The microbiome has many different functions. “One is to protect the human body against invading pathogens, whether they’re bacteria or viruses or fungi,” Blaser said. “That goes deep into evolution, maybe a billion years of evolution.”
Medical problems often arise when the balance between beneficial and pathogenic microbes in a person’s microbiome is thrown off, a condition known as dysbiosis.
The scientists studied microbiomes by measuring populations of microorganisms in stool samples taken from 60 subjects. The study group consisted of 20 COVID-19 patients, 20 healthy donors and 20 COVID-19-recovered subjects. They found major differences in the population numbers of 55 different species of bacteria when comparing the microbiomes of infected patients with the healthy and recovered patients.
The Rutgers scientists plan to continue to test and track the microbiomes of patients in the study to ascertain the long-term effect on individual microbiomes from COVID-19.
“Further investigation of patients will enhance understanding of the role of the gut microbiome in COVID-19 disease progression and recovery,” Blaser said. “These findings may help identify microbial targets and probiotic supplements for improving COVID-19 treatment.”
#functionalmedicine
#microbiome